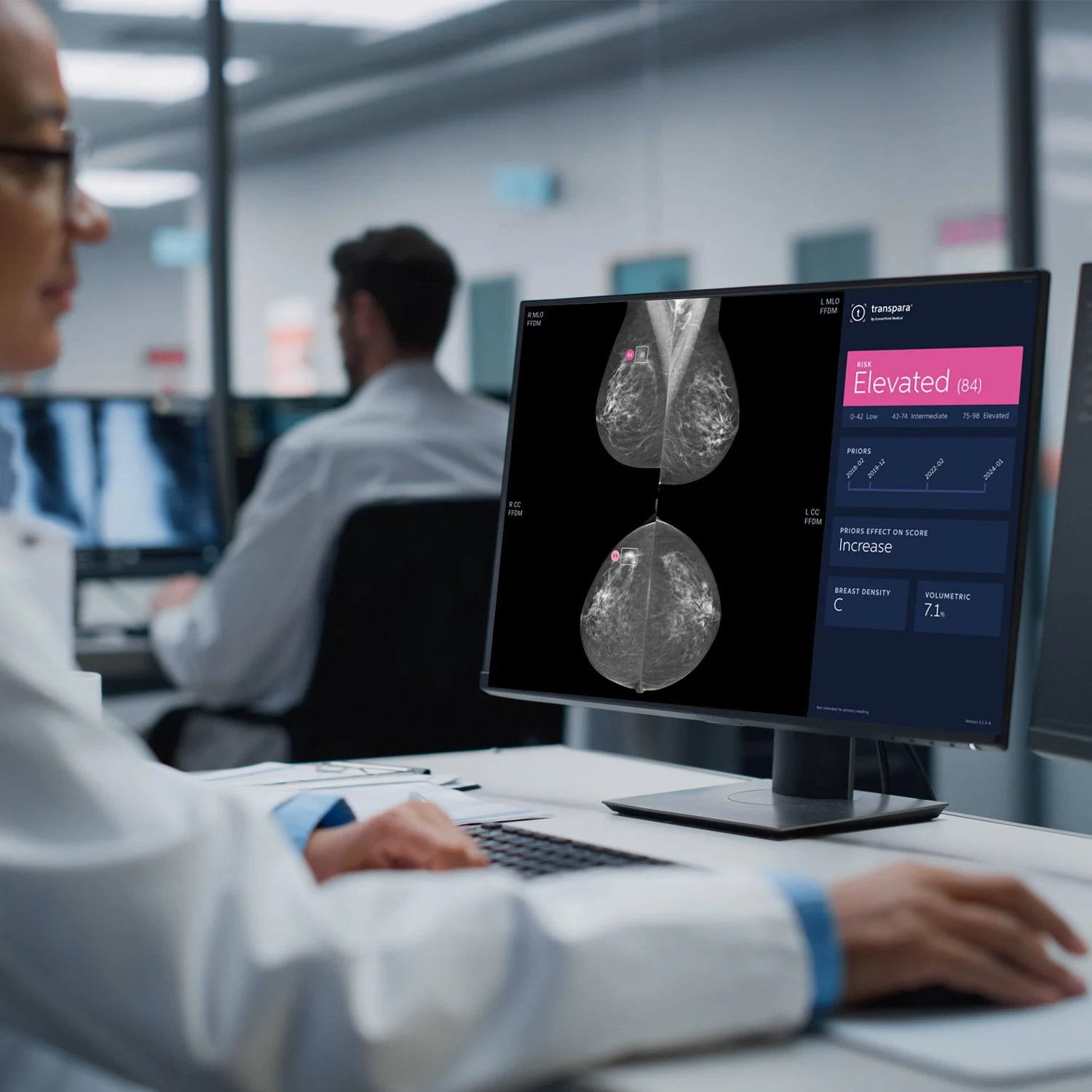
A large-scale clinical study conducted in Sweden as part of the national breast cancer screening program has yielded encouraging new results. Known as MASAI (Mammography Screening With Artificial Intelligence), this trial followed more than 105,000 women to evaluate the contribution of ScreenPoint's AI to mammograms. The study group was divided into two, with one group being monitored using Transpara Breast and the other using traditional methods. The research, led by Dr. Kristina Lång of Lund University (recognized many times by the European Society of Breast Imaging), shows that AI could improve the detection of breast cancer while reducing certain more aggressive cases detected between screenings.
The results of the MASAI trial are particularly striking. Thanks to the use of artificial intelligence, in addition to the expertise of radiologists, the detection rate for breast cancer has increased by 29%.
At the same time, cancers diagnosed between two screenings (interval cancers), which are often more difficult to treat, decreased by 12%. More specifically, the group using AI recorded 16% fewer invasive cancers, 21% fewer large tumors, and 27% fewer aggressive forms. These advances were achieved without increasing the number of false alarms. In summary, AI has made it possible to detect more relevant cancers earlier, while maintaining a level of reliability comparable to conventional screening.
"The MASAI trial shows that artificial intelligence applied to breast cancer detection has reached a decisive milestone: it helps healthcare professionals work more efficiently and, above all, it has a tangible impact on women around the world. Now that this level of evidence exists, the question is no longer whether AI should be used, but how to ensure that all women can benefit from it," says ScreenPoint CEO Pieter Kroese.
.webp)

.webp)
.webp)
.webp)

.webp)
.webp)




